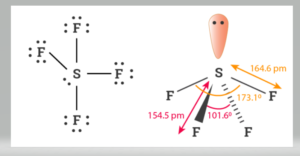
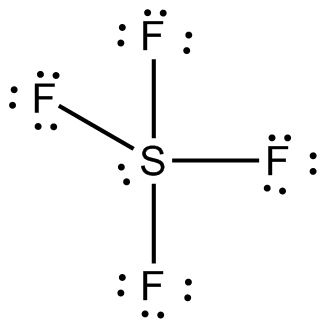
Molecular shapes of SF4, CF4 and XeF4 area)The same with 2, 0 and 1 lone pairs of electrons respectivelyb)The same, with 1, 1 and 1 lone pairs of electrons respectivelyc)Different, with 0,1

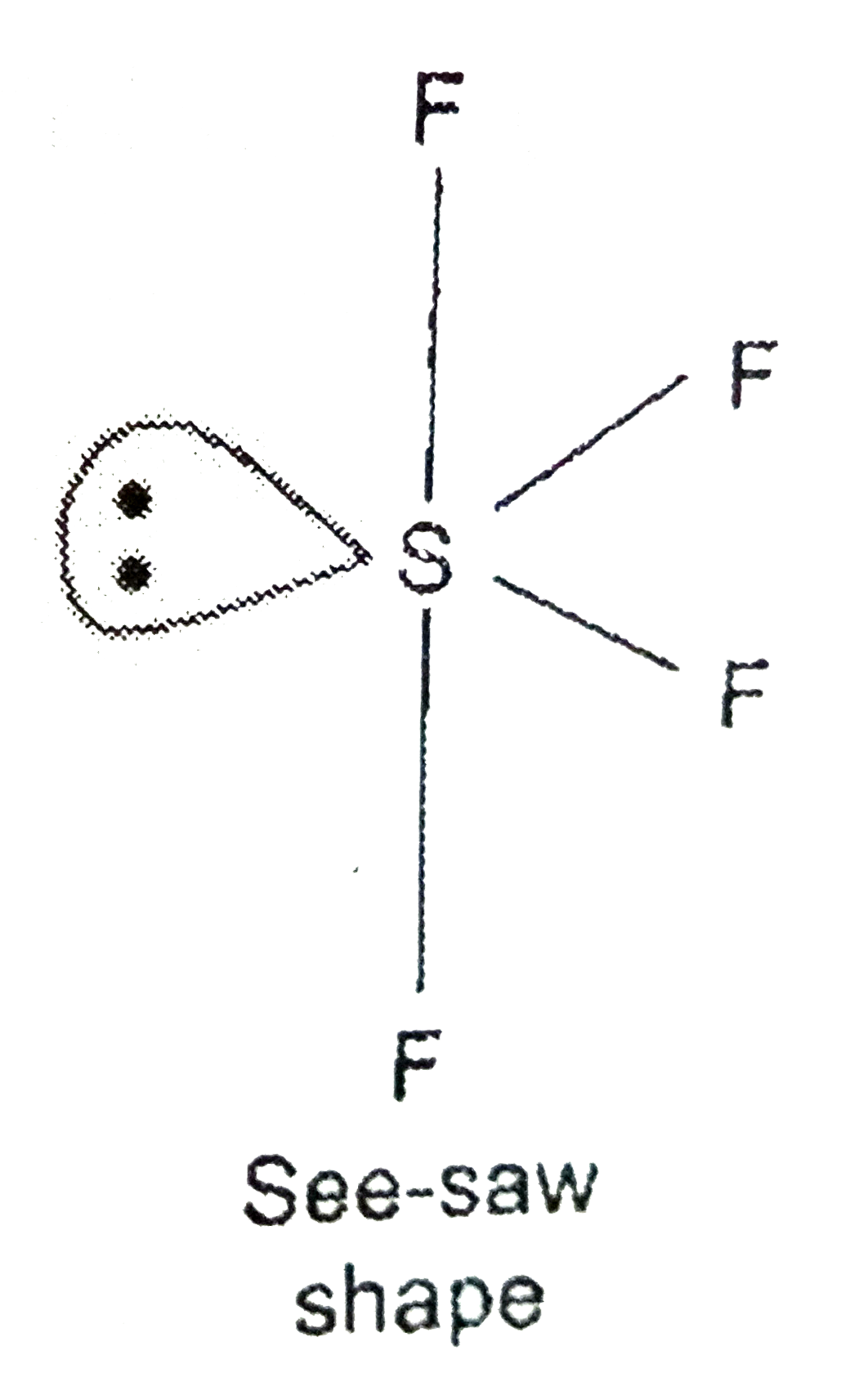
Draw the Lewis dot structure for SF4 and provide the following information. a. number of bond pairs b. number of lone pairs c. molecular geometry d. hybridization of the central atom

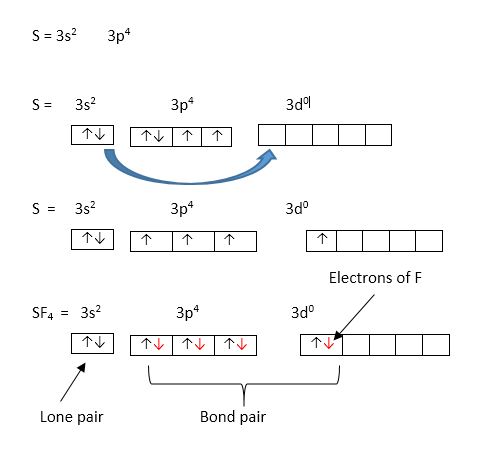
SOLVED: A. What is the hybridization of the central atom in SF4 Hybridization What are the approximate bond angles in this substance Bond angles What is the hybridization of the central atom
q a describe the hybridisation in case of pcl3 b decude the shape of sf4 molecule on basis of vsepr theory 0z73g4ww -Chemistry - TopperLearning.com

SOLVED: A. What is the hybridization of the central atom in SF4 Hybridization What are the approximate bond angles in this substance Bond angles What is the hybridization of the central atom