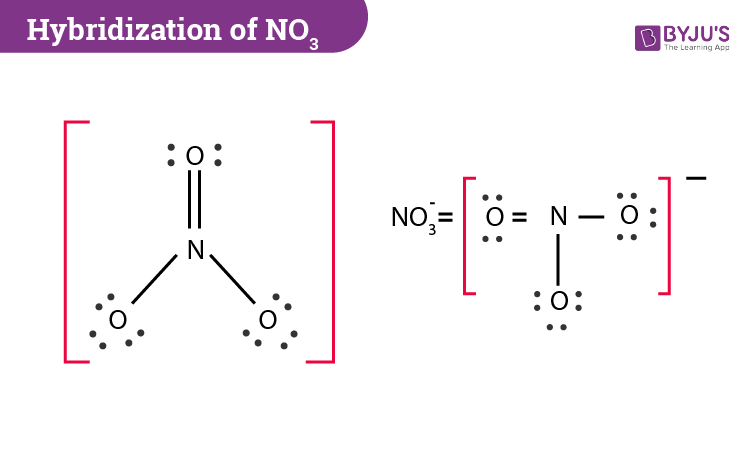
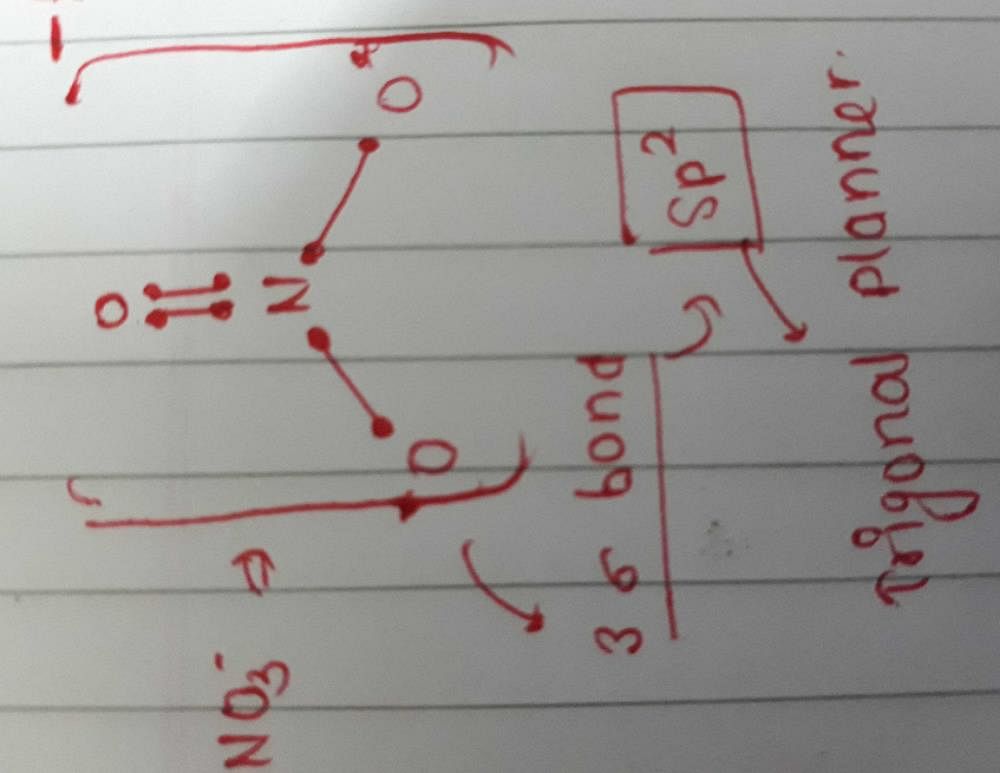
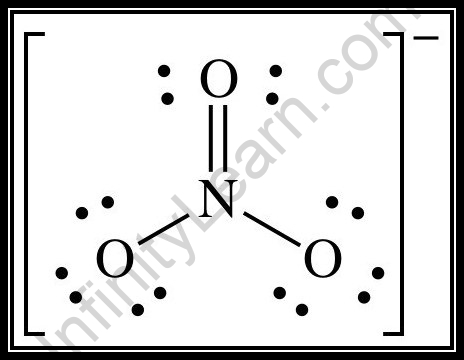
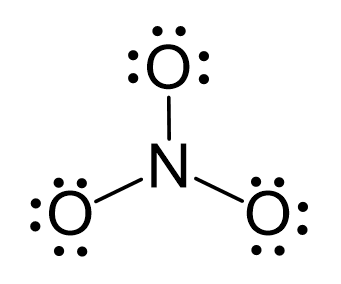
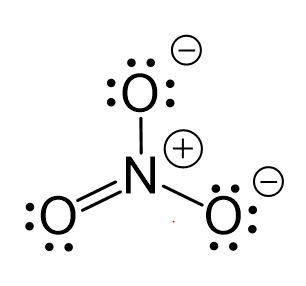
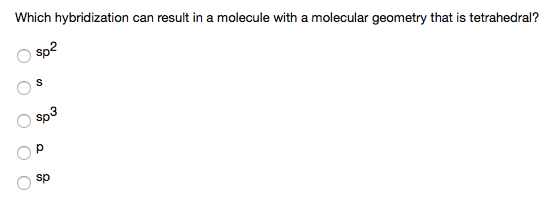SOLVED: Molecular Formula: nitrate anion NO3 Structure Molecular Geometry Valence Electron Around Each Atom Orbital Hybridization According to Bond Classifications Molecular Formula: carbonate anion CO3 Structure Electron Pair Geometry and Molecular ...
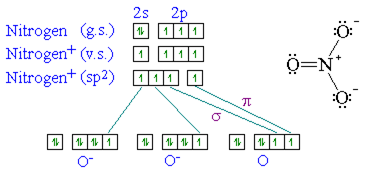
The hybridization of orbitals of N atom in NO3^- NO2^+ and NH4^+ are respectively - Sarthaks eConnect | Largest Online Education Community

NO3- lewis structure, molecular geometry, bond angle, hybridization | Molecular geometry, Molecular, Electron configuration

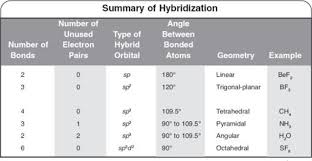
The hybridization of atomic orbital of nitrogen in NO2+, NO3- and NH4+ are:a)sp, sp2, sp3b)sp2, sp3, spc)sp2, sp, sp3d)sp, sp3, sp2Correct answer is option 'A'. Can you explain this answer? - EduRev